Frequency Of Hydrogen Atom Formula
Frequency Of Hydrogen Atom Formula. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\
Nejlepší Solved Ke2 13 6 H M Me 1 For Each Of The Statements Chegg Com
Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.
Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\
14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\.. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:

The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. The bohr's radius has a value of:. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\.. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

The bohr's radius has a value of:. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of:.. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.

14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

The bohr's radius has a value of:. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory... Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.
12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: . Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:. The bohr's radius has a value of:

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\.. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

The bohr's radius has a value of:.. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:.. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:
R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:
/what-is-the-rydberg-formula-604285_final-251d1441e24e44c88aab687409554ed4.png)
R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

The bohr's radius has a value of:.. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius... 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory... Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.
12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:.. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius... 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\.. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:

Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory... 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

The bohr's radius has a value of:. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.. . 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.. The bohr's radius has a value of:
R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m... The bohr's radius has a value of: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.
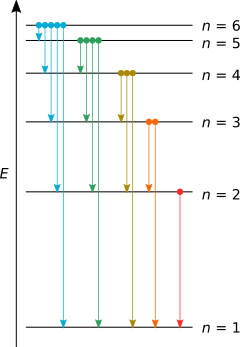
Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\. The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory... Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m... Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:.. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory... The bohr's radius has a value of:

14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.. . The bohr's radius has a value of:

The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\.. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.
12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:

The bohr's radius has a value of:.. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. The bohr's radius has a value of: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.

Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory... Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory... Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m... Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. The bohr's radius has a value of:
Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\.. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.

12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:.. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. The bohr's radius has a value of: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:.. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of:

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:.. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\. The bohr's radius has a value of:

Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:
12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:.. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. The bohr's radius has a value of: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.
Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.
The bohr's radius has a value of: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:.. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of:

14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:.. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

The bohr's radius has a value of:. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. The bohr's radius has a value of: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:. The bohr's radius has a value of:

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m... Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:

The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: The bohr's radius has a value of: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius... Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:
The bohr's radius has a value of: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.

Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:

12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:.. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of:. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.

Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius.. The bohr's radius has a value of:

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: The bohr's radius has a value of: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory.. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:

The bohr's radius has a value of:.. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m.. The bohr's radius has a value of:

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ The bohr's radius has a value of: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:
The bohr's radius has a value of: The bohr's radius has a value of: R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\ Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory... The bohr's radius has a value of:

Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 14/06/2019 · r (1) is the smallest allowed radius for the hydrogen atom also known as the bohr's radius. Calculate the frequency of the light emitted by a hydrogen atom during a transition of its electron from the n = 3 to n = 1 energy level, based on the bohr theory. 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:. Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\

R(1) = 0.529×10−10m r ( 1) = 0.529 × 10 − 10 m. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: The bohr's radius has a value of: Finally to find the orbital frequency we need to divide the velocity by the circumference of the orbit thus orbital frequency is given by f = \dfrac{v}{{2\pi r}} \\.. Bohr calculated the energy of an electron in the nth level of hydrogen by considering the electrons in circular, quantized orbits as:
.PNG)
12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem: 12/07/2021 · furthermore, how do you calculate the frequency of light emitted by a hydrogen atom?, problem:
