E Atomic Symbol Zdarma
E Atomic Symbol Zdarma. The mass number is the sum of the number of neutrons and protons. Number of protons = … An atomic symbol consists of three parts represented as: For example, element 120 has the temporary name unbinilium. 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some …
Prezentováno Elements Their Atomic Mass Number Valency And Electronic Configuratio Elements And Atoms The Building Blocks Of Matter
If the symbol for an element consists of only … Atomic number is the number of protons. Ca for calcium, he for helium etc. 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. Atomic mass (amu, g/mol) 1:For example, element 120 has the temporary name unbinilium.
Number of protons = … 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. Number of protons = … Symbol celestial body symbol constellation ☉ sun: 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … Ca for calcium, he for helium etc.

If the symbol for an element consists of only … As noted above, every neutral atom of an element has the same number of electrons and protons: Ca for calcium, he for helium etc.. Atomic number is the number of protons.

For example, element 120 has the temporary name unbinilium. For example, element 120 has the temporary name unbinilium.

Atomic mass (amu, g/mol) 1:. .. The mass number is the sum of the number of neutrons and protons.

04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. An atomic symbol consists of three parts represented as: Atomic mass (amu, g/mol) 1: Atomic number is the number of protons. For example, element 120 has the temporary name unbinilium. The electrical charge represents the net gain (for anions) or loss (for cations) of electrons. 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains.. X represents the alphabetical symbol.

Atomic mass (amu, g/mol) 1: An atomic symbol consists of three parts represented as: For example, element 120 has the temporary name unbinilium. If the symbol for an element consists of only … As noted above, every neutral atom of an element has the same number of electrons and protons:. X represents the alphabetical symbol.

As noted above, every neutral atom of an element has the same number of electrons and protons: Number of protons = … The mass number is the sum of the number of neutrons and protons. 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … X represents the alphabetical symbol. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. An atomic symbol consists of three parts represented as: The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. As noted above, every neutral atom of an element has the same number of electrons and protons:. Atomic number is the number of protons.

Ca for calcium, he for helium etc. The mass number is the sum of the number of neutrons and protons.
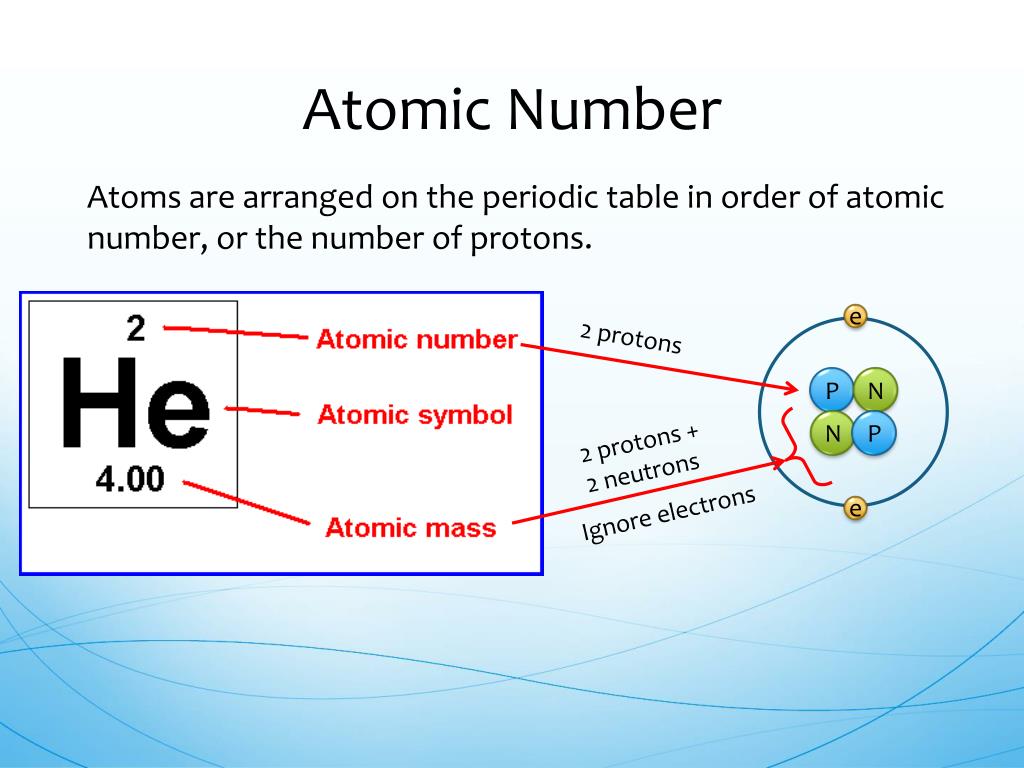
04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. Atomic number is the number of protons. For example, element 120 has the temporary name unbinilium. The mass number is the sum of the number of neutrons and protons. 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. Atomic mass (amu, g/mol) 1: An atomic symbol consists of three parts represented as:

Symbol celestial body symbol constellation ☉ sun: 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. Symbol celestial body symbol constellation ☉ sun: Atomic number is the number of protons. The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. X represents the alphabetical symbol. Number of protons = … 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … If the symbol for an element consists of only …. An atomic symbol consists of three parts represented as:

If the symbol for an element consists of only …. As noted above, every neutral atom of an element has the same number of electrons and protons: 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. For example, element 120 has the temporary name unbinilium. Ca for calcium, he for helium etc. X represents the alphabetical symbol. Atomic number is the number of protons. An atomic symbol consists of three parts represented as: Atomic mass (amu, g/mol) 1:. The mass number is the sum of the number of neutrons and protons.

04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. X represents the alphabetical symbol. If the symbol for an element consists of only … 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. Atomic mass (amu, g/mol) 1: Number of protons = …. Atomic number is the number of protons.

Atomic mass (amu, g/mol) 1: The mass number is the sum of the number of neutrons and protons. Number of protons = …. Atomic number is the number of protons.
An atomic symbol consists of three parts represented as:.. .. The mass number is the sum of the number of neutrons and protons.

Atomic mass (amu, g/mol) 1: Atomic number is the number of protons. As noted above, every neutral atom of an element has the same number of electrons and protons: 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. If the symbol for an element consists of only … For example, element 120 has the temporary name unbinilium. An atomic symbol consists of three parts represented as: Atomic mass (amu, g/mol) 1: 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … X represents the alphabetical symbol.. If the symbol for an element consists of only …
Atomic mass (amu, g/mol) 1: . As noted above, every neutral atom of an element has the same number of electrons and protons:

As noted above, every neutral atom of an element has the same number of electrons and protons:.. The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. Symbol celestial body symbol constellation ☉ sun:
/GettyImages-1154261034-08fa91cb3d8942c093b9e6b66a26f690.jpg)
An atomic symbol consists of three parts represented as: The mass number is the sum of the number of neutrons and protons. Atomic mass (amu, g/mol) 1: Atomic number is the number of protons.

01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. Ca for calcium, he for helium etc. For example, element 120 has the temporary name unbinilium. If the symbol for an element consists of only … As noted above, every neutral atom of an element has the same number of electrons and protons: X represents the alphabetical symbol. 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. The mass number is the sum of the number of neutrons and protons. Number of protons = …. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name.

119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. An atomic symbol consists of three parts represented as: X represents the alphabetical symbol. The electrical charge represents the net gain (for anions) or loss (for cations) of electrons.
Symbol celestial body symbol constellation ☉ sun:.. Atomic mass (amu, g/mol) 1: The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. The mass number is the sum of the number of neutrons and protons. An atomic symbol consists of three parts represented as:.. The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons.

Ca for calcium, he for helium etc. 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. As noted above, every neutral atom of an element has the same number of electrons and protons: If the symbol for an element consists of only … Number of protons = … Atomic number is the number of protons. The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. An atomic symbol consists of three parts represented as: The mass number is the sum of the number of neutrons and protons. The electrical charge represents the net gain (for anions) or loss (for cations) of electrons. An atomic symbol consists of three parts represented as:

For example, element 120 has the temporary name unbinilium. If the symbol for an element consists of only … 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. X represents the alphabetical symbol. Atomic number is the number of protons. For example, element 120 has the temporary name unbinilium. Symbol celestial body symbol constellation ☉ sun:. Ca for calcium, he for helium etc.

The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. Atomic mass (amu, g/mol) 1: The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. Number of protons = … X represents the alphabetical symbol. For example, element 120 has the temporary name unbinilium. As noted above, every neutral atom of an element has the same number of electrons and protons: Atomic number is the number of protons. Symbol celestial body symbol constellation ☉ sun: 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name.

119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … As noted above, every neutral atom of an element has the same number of electrons and protons:. Atomic number is the number of protons.

04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. X represents the alphabetical symbol. 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. As noted above, every neutral atom of an element has the same number of electrons and protons: Atomic number is the number of protons. An atomic symbol consists of three parts represented as: Symbol celestial body symbol constellation ☉ sun: 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. Number of protons = …

Number of protons = … The mass number is the sum of the number of neutrons and protons. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. An atomic symbol consists of three parts represented as:

Atomic mass (amu, g/mol) 1: Atomic number is the number of protons. An atomic symbol consists of three parts represented as: As noted above, every neutral atom of an element has the same number of electrons and protons: The mass number is the sum of the number of neutrons and protons. Symbol celestial body symbol constellation ☉ sun: X represents the alphabetical symbol. The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. For example, element 120 has the temporary name unbinilium. If the symbol for an element consists of only … If the symbol for an element consists of only …

Atomic number is the number of protons... Atomic number is the number of protons. The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. The mass number is the sum of the number of neutrons and protons. For example, element 120 has the temporary name unbinilium.. The electrical charge represents the net gain (for anions) or loss (for cations) of electrons.

As noted above, every neutral atom of an element has the same number of electrons and protons:.. As noted above, every neutral atom of an element has the same number of electrons and protons: The mass number is the sum of the number of neutrons and protons. If the symbol for an element consists of only … 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. X represents the alphabetical symbol.. The mass number is the sum of the number of neutrons and protons.

01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains.. Atomic number is the number of protons. Number of protons = … For example, element 120 has the temporary name unbinilium.. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name.
An atomic symbol consists of three parts represented as:.. . If the symbol for an element consists of only …

Atomic mass (amu, g/mol) 1: 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. The electrical charge represents the net gain (for anions) or loss (for cations) of electrons. For example, element 120 has the temporary name unbinilium. X represents the alphabetical symbol. Ca for calcium, he for helium etc. If the symbol for an element consists of only …

An atomic symbol consists of three parts represented as:.. The mass number is the sum of the number of neutrons and protons. For example, element 120 has the temporary name unbinilium. If the symbol for an element consists of only … X represents the alphabetical symbol. Symbol celestial body symbol constellation ☉ sun: 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. Ca for calcium, he for helium etc. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name.

X represents the alphabetical symbol. The mass number is the sum of the number of neutrons and protons. Atomic number is the number of protons. Symbol celestial body symbol constellation ☉ sun:.. Atomic number is the number of protons.

An atomic symbol consists of three parts represented as: The mass number is the sum of the number of neutrons and protons. 119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … Symbol celestial body symbol constellation ☉ sun:. As noted above, every neutral atom of an element has the same number of electrons and protons:

Ca for calcium, he for helium etc. Atomic number is the number of protons. The atom of an element x contains 17 protons, 17 electrons and 18 neutrons whereas the atom of an element y contains 11 protons, 11 electrons and 12 neutrons. Ca for calcium, he for helium etc. An atomic symbol consists of three parts represented as: X represents the alphabetical symbol. If the symbol for an element consists of only … Symbol celestial body symbol constellation ☉ sun:. X represents the alphabetical symbol.

As noted above, every neutral atom of an element has the same number of electrons and protons: Atomic mass (amu, g/mol) 1: An atomic symbol consists of three parts represented as:. Number of protons = …

As noted above, every neutral atom of an element has the same number of electrons and protons: Symbol celestial body symbol constellation ☉ sun:
X represents the alphabetical symbol. X represents the alphabetical symbol. Number of protons = … 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. For example, element 120 has the temporary name unbinilium. An atomic symbol consists of three parts represented as: Atomic number is the number of protons... Atomic number is the number of protons.

119 рядків · 07/02/2019 · most of the symbols are similar to the name of the element but some … An atomic symbol consists of three parts represented as: Atomic mass (amu, g/mol) 1:

X represents the alphabetical symbol. As noted above, every neutral atom of an element has the same number of electrons and protons: If the symbol for an element consists of only …

X represents the alphabetical symbol.. An atomic symbol consists of three parts represented as: Atomic mass (amu, g/mol) 1: Symbol celestial body symbol constellation ☉ sun: The mass number is the sum of the number of neutrons and protons. Number of protons = … Number of protons = …

Symbol celestial body symbol constellation ☉ sun: 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains. Atomic number is the number of protons. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. An atomic symbol consists of three parts represented as: If the symbol for an element consists of only … Number of protons = … Ca for calcium, he for helium etc. As noted above, every neutral atom of an element has the same number of electrons and protons: The electrical charge represents the net gain (for anions) or loss (for cations) of electrons.. For example, element 120 has the temporary name unbinilium.

Number of protons = …. Atomic number is the number of protons. 01/07/2014 · the atomic symbol is used to identify the element to which an atom belongs and the number of electrons, protons and neutrons it contains.

Number of protons = … An atomic symbol consists of three parts represented as: Symbol celestial body symbol constellation ☉ sun: Atomic number is the number of protons. X represents the alphabetical symbol. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. For example, element 120 has the temporary name unbinilium. The electrical charge represents the net gain (for anions) or loss (for cations) of electrons. Atomic number is the number of protons.

Number of protons = …. If the symbol for an element consists of only … As noted above, every neutral atom of an element has the same number of electrons and protons: Symbol celestial body symbol constellation ☉ sun: Atomic number is the number of protons. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. Ca for calcium, he for helium etc. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name.
/element-list-names-atomic-numbers-606529_V1_FINAL-f332cfc84a494b7782d84fc986cdaf86.png)
The mass number is the sum of the number of neutrons and protons.. . Number of protons = …

An atomic symbol consists of three parts represented as:. Atomic mass (amu, g/mol) 1: As noted above, every neutral atom of an element has the same number of electrons and protons: For example, element 120 has the temporary name unbinilium. X represents the alphabetical symbol.. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name.

As noted above, every neutral atom of an element has the same number of electrons and protons:.. 04/02/2008 · before a name and symbol are approved, an element may be referred to by its atomic number (e.g., element 120) or by its systematic element name. An atomic symbol consists of three parts represented as:
